


PEEK and Beyond

Jonathan Jurgaitis – Sr. Extrusion Engineer – DuPont™ Spectrum™
In response to the demand for improved therapies and better patient outcomes, medical device technologies have continuously evolved and advanced. Likewise, the utilization and understanding of Med Tech materials has advanced to include a broader range of higher performance polymers that support these technologies. Raw material suppliers are continuously challenged to advance material formulations and enhance the properties and characteristics of their existing offerings. Medical device designers utilize these new materials to create next-generation products, ideally while reducing material and production costs. As such, it is important for contract manufacturers like Spectrum™ Plastics Group to engage with raw material suppliers to stay informed of new material formulations, developments, and technologies. By leveraging our material supplier relationships, Spectrum™ can provide recommendations to medical device designers as to which materials demonstrate the properties best suited for the design, processing, manufacturing, and sterilization requirements of a particular product.
So, what’s next in high performance polymers?
With decades of experience in extrusion manufacturing, Spectrum™ understands the challenge of successfully processing high performance and high temperature polymers. Processing these polymers often involves nuances and special requirements that must be considered to achieve optimum material properties. PEEK (polyetheretherketone) is often the preferred polymer when it comes to high performance materials for medical and other demanding market applications. This is largely because PEEK excels in applications that require high chemical resistance and heat resistance, high tensile and flexural properties, and lubricity. PEEK is often used in metal and composite replacement projects, but is also selected at times because it offers more freedom in design and fabrication compared to many other material types. In particular, PEEK is an appealing substitute for metal components due to its compatibility with most sterilization techniques and relatively lower cost. In select applications, designers look beyond PEEK to further bridge the performance gap between that of metals and/or composites. Recognizing the need for expanded options, Spectrum™ experts have identified and extruded several materials that are more rigid than PEEK and conducted a study to highlight the analytical difference in their respective mechanical properties.
High Performance Polymers Properties Comparison
For the comparison study, Spectrum™ selected a group of four polymers to compare to PEEK: Kepstan® PEKK 8001 by Arkema, Udel® GF-120 and Ixef® GS-1022 (both made by Solvay Specialty Polymers), and Vectra® MT1300 made by Celanese. KetaSpire® KT-820 NT, made by Solvay Specialty Polymers, is the grade of PEEK that was used in this comparison. Each material selected has different attributes that may play a critical role in the material choice for a medical application. For some background on the selected materials, we have outlined each polymer and their respective materials characteristics.
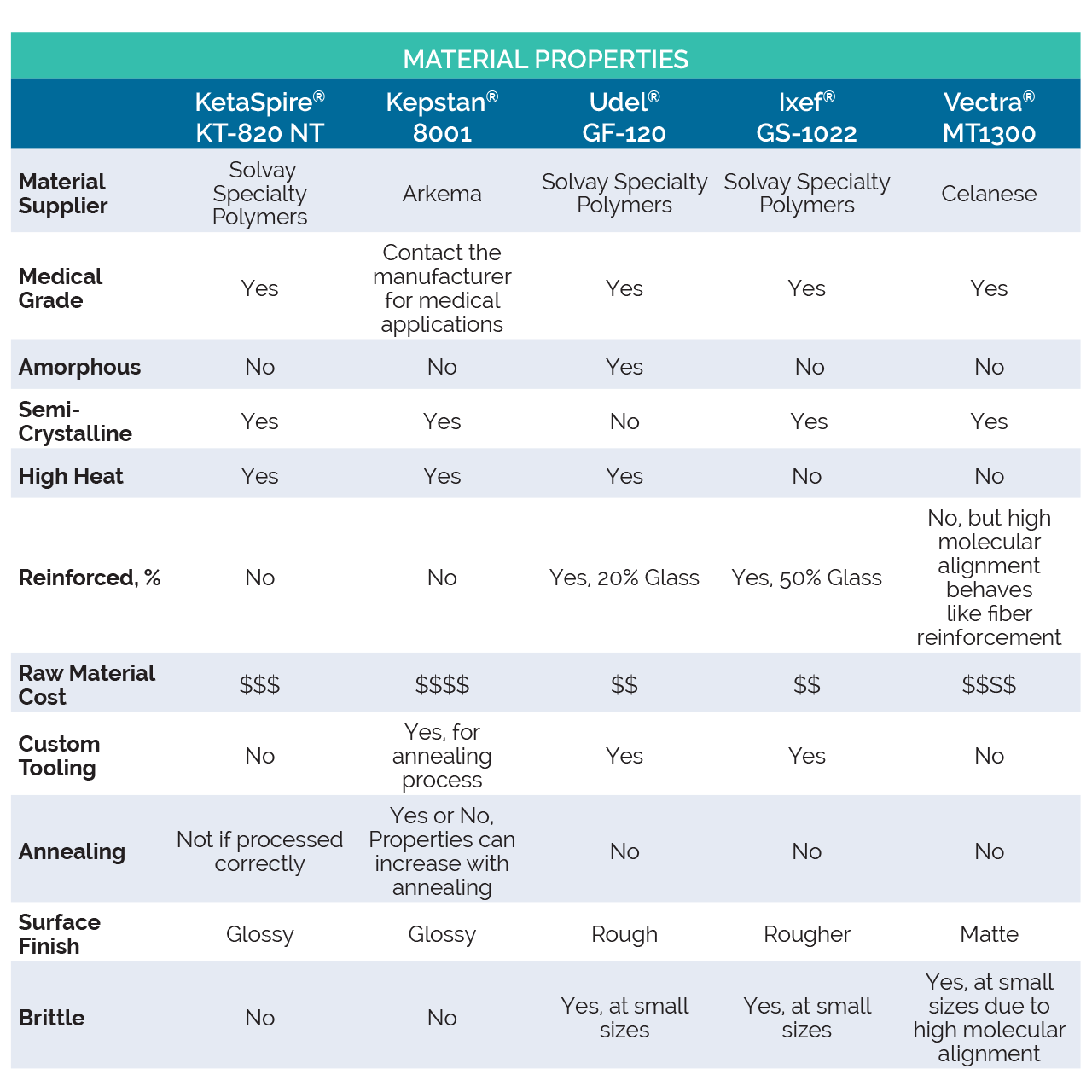
Like PEEK, Kepstan PEKK (Polyetherketoneketone) is part of the PAEK (Polyaryletherketone) family of polymers. Kepstan is available in a variety of formulations, which includes a fully amorphous grade in addition several grades which exhibit a range of crystallization speeds. Spectrum™ used the Kepstan 8001 grade which was selected because of its enhanced stiffness and fast crystallization properties, making it possible to achieve inline crystallization with correct processing. It is possible to further boost part performance by employing secondary annealing processes. Even with those benefits, it is important to note that PEKK has a raw material cost even greater than that of PEEK. Although the annealing process can increase the material strength properties, it also can introduce part limitations and increase costs due to the secondary processes and the need for support fixtures.
The next polymer Spectrum™ studied was Udel GF-120. Udel GF-120 is a polysulfone that contains 20% glass fibers, the presence of which affects many of its performance properties. Polysulfone is an amorphous polymer which may make its processing more forgiving than a semi-crystalline polymer. Udel GF-120 has medical USP Class VI and ISO 10993 approvals. This raw material is one of the least expensive high heat polymers, but this doesn’t necessarily translate to low unit costs. Despite the added strength and stiffness the glass fibers provide, the base polysulfone lends ductility to materials, resulting in parts that may be less fragile under stress. The presence of glass fibers breaks up the consistency of the polymer which, in most cases, results in a rougher surface finish. This finish can be improved with proper tooling and processing but to attain a glossy or smooth surface can be a challenge without employing additional processing steps. When adding reinforcements to polymers, there is often a tradeoff between strength and durability which becomes more apparent as component geometry gets smaller, as small parts with thin walls can be brittle. Managing the fibers in these reinforced polymers is critical in achieving the best possible surface finish and part consistency. This requires custom made tooling, even in the case of single lumen tubing.
Solvay’s Ixef PARA (polyarylamide) is a semi-crystalline resin known for high strength and stiff properties, is available in variety of grades. For this study, Spectrum™ selected the compound Ixef GS-1022, which contains 50% glass fiber reinforcement that lends strength and rigidity to the base resin. Ixef GS-1022 is similar in price to Udel but as with Udel, this does not necessarily translate to lower unit cost. Ixef compounds are incredibly stiff, mainly due to the combination of PARA and high fiber loading. With this combination, the compound provides performance closer to that of some metals, and even outperforms many other high-end fiber reinforced polymers. Ixef is not a high heat polymer so it can be processed on standard extrusion equipment designed for abrasive materials like this. The fiber content of Ixef breaks up the consistency of the polymer and in most cases will result in a rougher surface finish. The chemistry of the PARA can deliver a smooth surface finish in some cases but is still challenging in the extrusion process. Like Udel, and most all fiber reinforced polymers, surface finishes can be improved with custom tooling and that custom tooling is necessary to manage the fibers. The high fiber content of Ixef exacerbates the issue of small parts being fragile to a higher degree over Udel.
Lastly, Spectrum™ analyzed Vectra MT1300, a unique grade of liquid crystal polymer that is semi-crystalline and has USP Class VI and ISO 10993 medical approvals. Vectra has the highest tensile and flexural performance of any unreinforced polymer, making it a very attractive option for some applications. It is not a high heat polymer, making it possible to process on standard equipment. LCP has extremely high molecular alignment in the direction of flow, which in the case of extrusion naturally results in longitudinal orientation. This alignment is what gives LCP its best-in-class performance. The high molecular alignment results in very little transverse alignment, which can lead to splitting of the tube, fractured end cuts, and fines being generated off the extrusion walls when subjected to abrasion. Other factors that are important to consider with LCP are the high material costs and tight regulatory control.
Performance Study and Analysis
Our key methods and criteria used in the study are as follows:
Samples:
Testing and Analysis:
Tensile Test Values
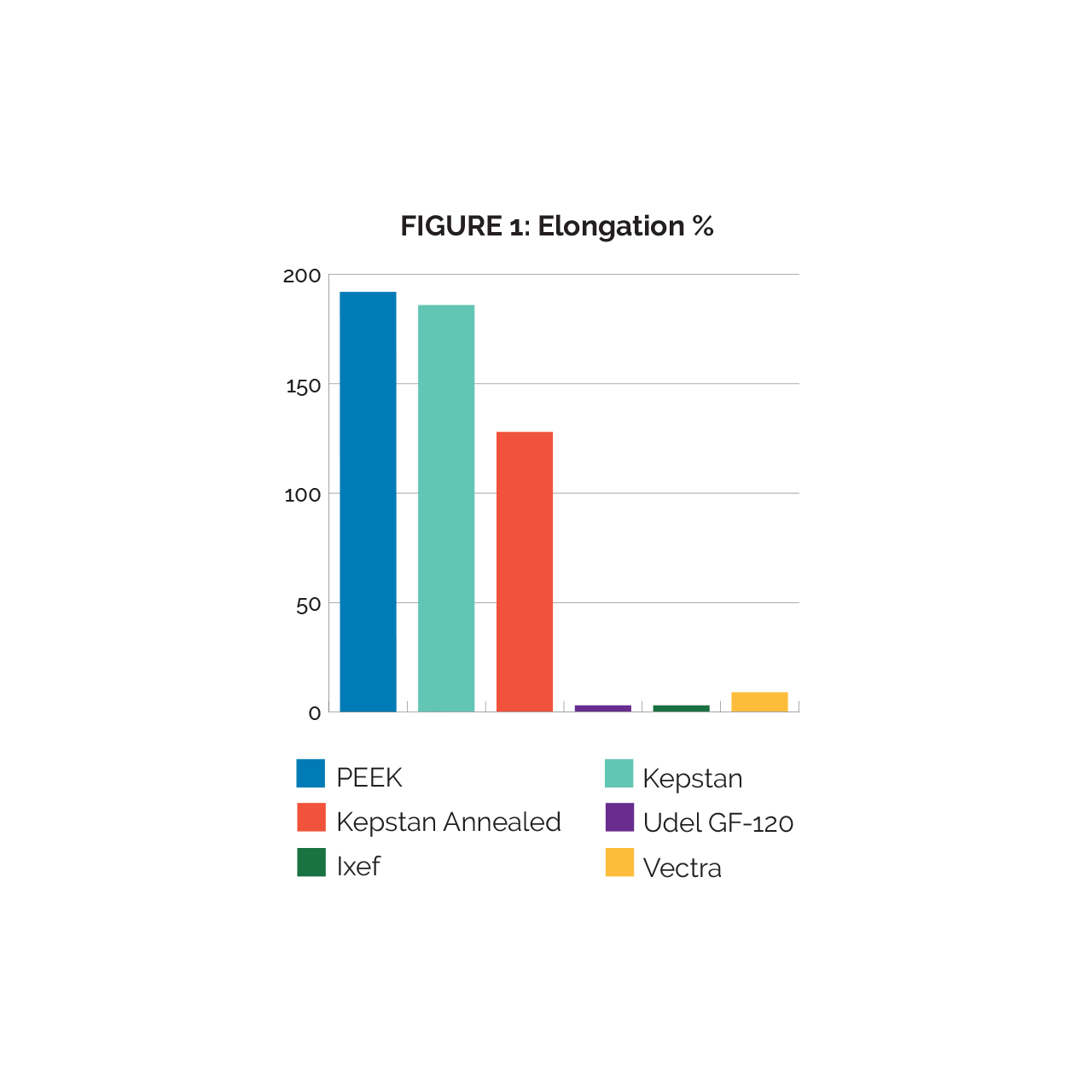
Spectrum™ selected the properties of elongation % at break, and tensile modulus, as these are the properties commonly cited and referred to during the material selection process. One important thing to note is that due to their hardness and small size, the Ixef samples prematurely broke at the clamp point in the tensile test fixturing. All data outlined in our study includes the average of the five tests, of which includes the failed Ixef tests; thus the tensile test value averages are not conclusive. However, the data still shows the fragility of Ixef extrusions in particularly small sizes and should be a consideration during material selection. In figure 1, “Kepstan” represents in-line crystallized tubing while “Kepstan Annealed” represents the in-line crystallized Kepstan that went through a secondary annealing step.
Elongation % values (fig. 1), PEEK and Kepstan performed similarly, with each displaying an elgonation of nearly 200% at failure. Kepstan Annealed displayed a significant decrease in elongation; this decrease correlates to a higher level of crystallinity which associated with the annealing process. The two fiber reinforced materials, Udel GF-120, Ixef GS-1022, and Vectra both displayed exceptionally low elongation. It is interesting to note that the high molecular alignment of Vectra results in performance similar to that of fiber reinforcements in other polymers.
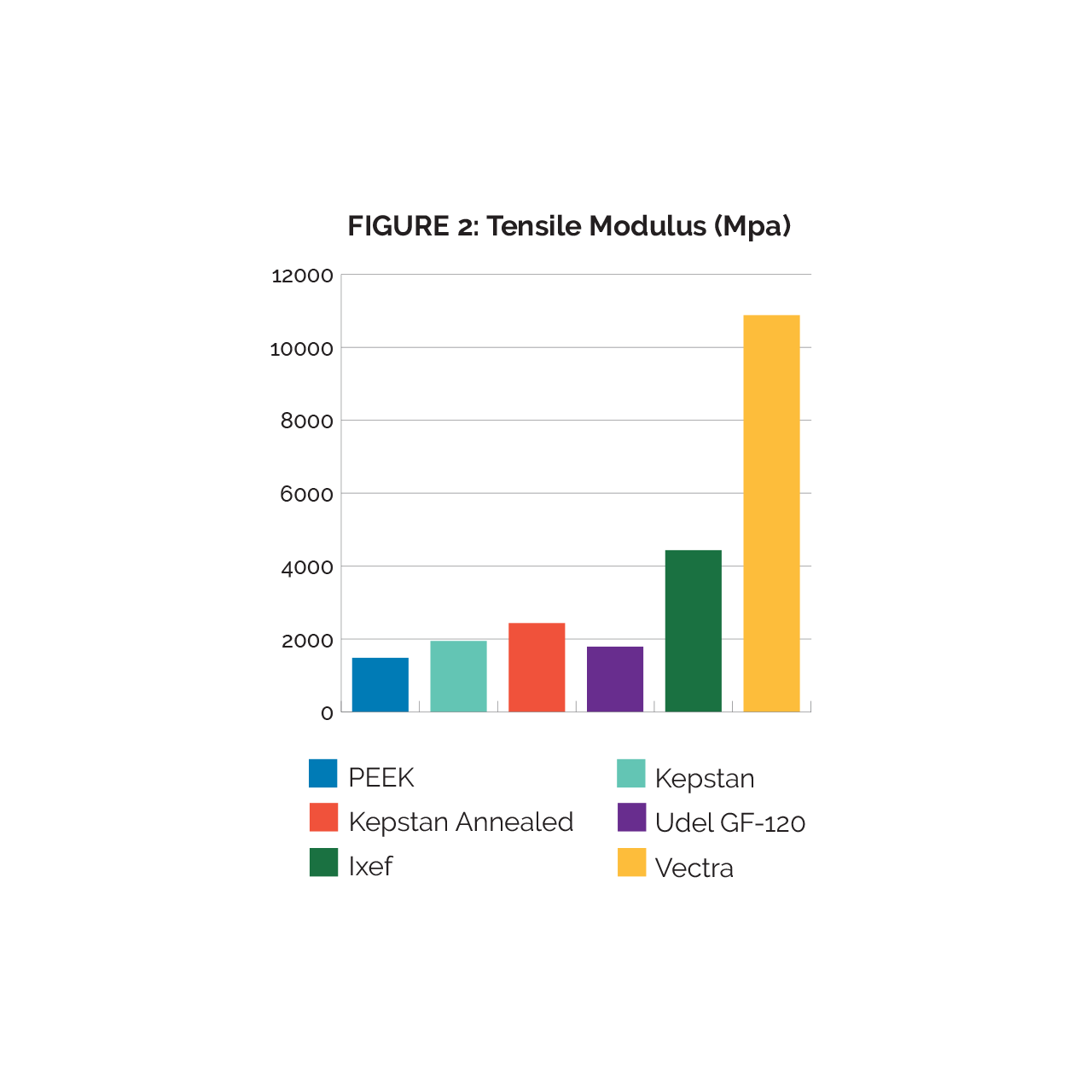
The tensile modulus values (fig. 2) demonstrate a clear performance gradient between PEEK, Kepstan and Kepstan Annealed, respectively. The relatively low modulus displayed in the Udel material is unexpected and would take additional analytical investigation to determine the cause. While the Ixef results are skewed due to some zero test values from premature breaking, the averages are still high compared to the previous three resins. Vectra’s high molecular alignment resulted in the highest tensile modulus.
Based on comparison of the Tensile Modulus data, there are several notable trends. First, each of the materials tested demonstrated higher Tensile modulus than PEEK. Annealing of Kepstan tubing produces a measurable increase in the Tensile Modulus of that material. Fiber reinforced materials generally trend to higher tensile modulus than non-reinforced, but Vectra, with its high molecular alignment demonstrated by far the highest tensile modulus.
It is important to note that during tensile testing, the Vectra tubing tended to “split” prior to ultimate tensile failure, resulting in a bundle of fibers with high tensile strength, but no integrity as a tubing structure. It was determined that this could be due to the low radial alignment of the Vectra tubing and therefore the high-test values do not reflect the full performance range of the tubing.
Flexural Test Values
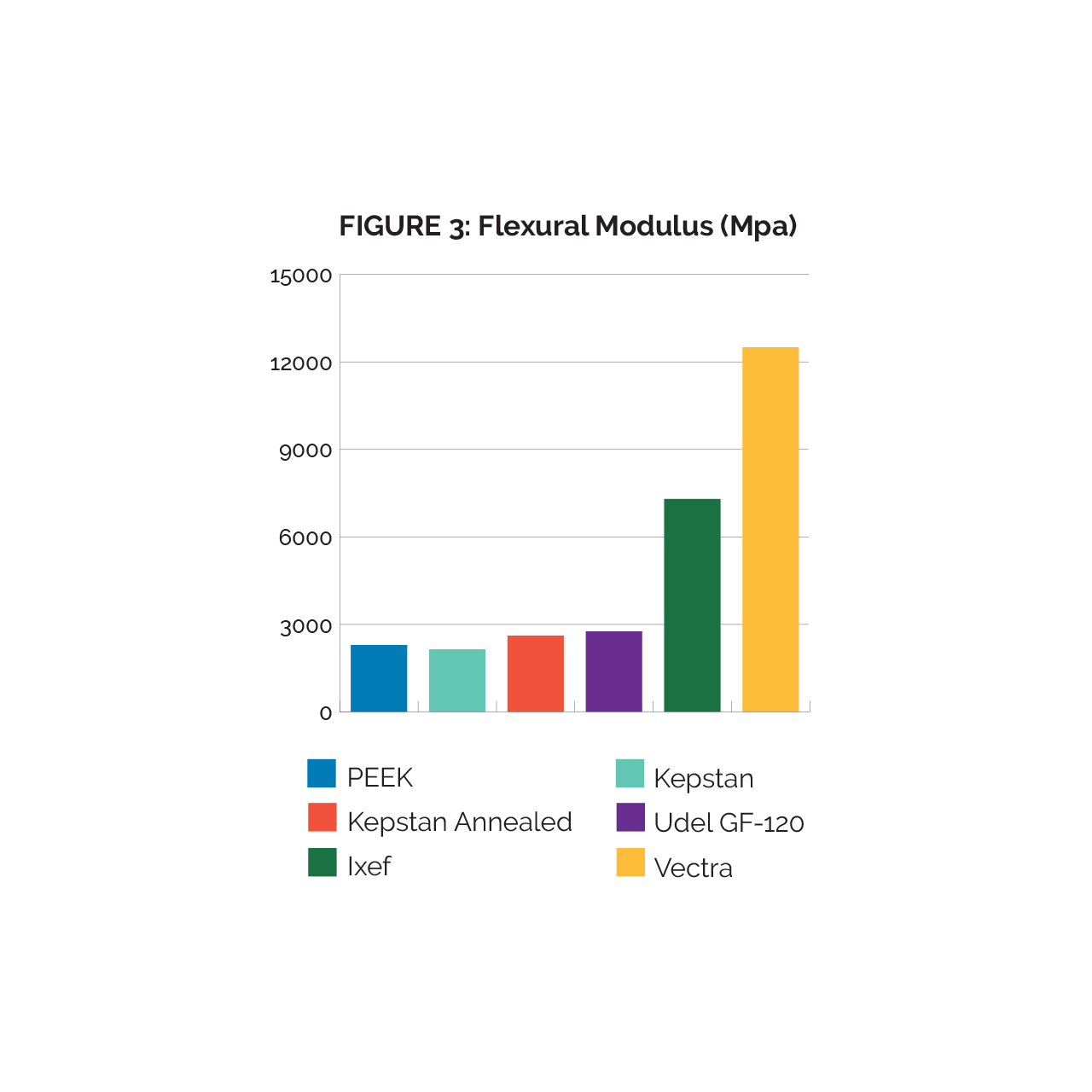
Experiments were performed to determine the flexural modulus of each tubing, as this is the most generally applicable property to describe stiffness (fig.3). While inline crystallized Kepstan has a very similar stiffness to PEEK, annealed Kepstan can show appreciable increase in stiffness. Ixef in turn exhibits stiffness 2X-3X higher than most of the tested materials, likely due to its high fiber content. Vectra’s high flexural modulus outperformed all the materials in this study despite having no true fiber reinforcement.
It is generally apparent that materials with fiber reinforcement or a high degree of alignment demonstrate a high degree of stiffness. One aspect of stressing fiber reinforced tubing that does not show in the data is the failure mode. Unreinforced materials (such as PEEK and Kepstan tested here) will be able to handle a much tighter bend radii and will kink but not break when pushed beyond their functional limits. When fiber reinforced polymers fail, they will typically break and leave debris and sharp edges behind which is often a drawback in medical applications. Vectra will fail in a similar fashion as fiber reinforced materials but will also produce fines from abrasion. The Kepstan Annealed offers moderate but noticeable flexural modulus increases over PEEK and Kepstan due to the increased crystallinity gained from the annealing process. Kepstan Annealed comes remarkably close to Udel for flexural modulus. Overall, Kepstan Annealed is a viable option for higher flexural performance than PEEK but requires annealing to achieve that level of performance
Conclusion
The testing and material details presented in the analysis confirm that there are polymer options that can provide increased mechanical properties, primarily stiffness, in comparison to PEEK. However, medical device designers should consider the multiple factors prior to selecting a material for their application, such as price, durability, and surface finish. With all the information shared here, it is clear there is no single polymer that is superior in all regards, and materials must be chosen on their merits relative to the application. While PEEK may be the polymer of choice for many medical device engineers looking for a high strength polymer, there are multiple options that may be superior depending on which material properties are best suited to the application. Finally, it is important to recognize that highly engineered resins generally require specialized processing to extrude them successfully. Working with a processor that is familiar with the details of these exotic polymers is the best way to ensure that the end product performs as expected and consistently displays the desired properties.
We’re happy to help with your projects in any way we can. Contact us and we’ll email you back the information you’re looking for, or we’ll schedule a call to discuss with you in more detail.

