
Multishot injection molding requires expertise in material interactions and understanding of how to combine multiple materials in one mold. We have the capability to support complex components from concept to production.
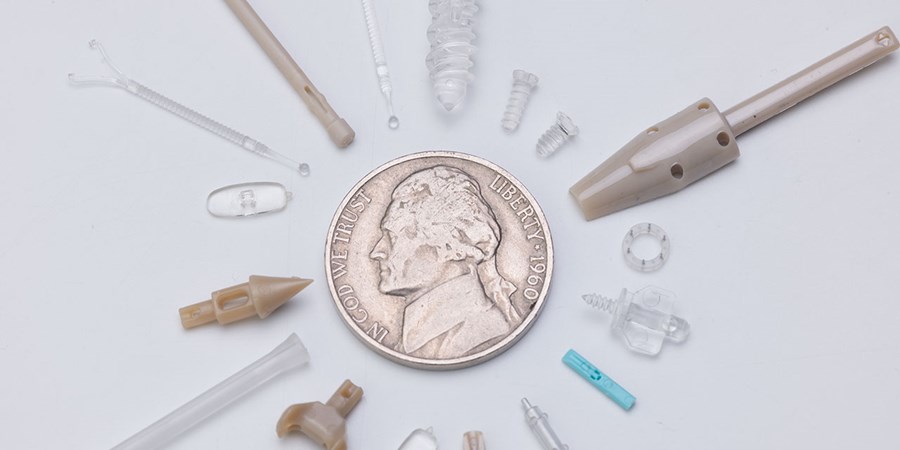
As medical treatments become less invasive, medical devices are getting smaller. We have the expertise to do precision micromolding, including specialized equipment, tool designs, and measurement systems.

We have decades of experience molding onto metal and other substrates. Combining properties of different materials, such as strength and flexibility, can create more intricate designs for aesthetics, branding, or ergonomics.

Implantable and bioresorbable materials pose unique preparation, processing, and handling challenges. As a pioneer with these materials, we have expertise and dedicated resources for manufacturing implantable and bioresorbable plastic components.

Our silicone molding capabilities include liquid injection molding as well as silicone rubber transfer molding, overmolding, and liquid injection molding.

As a single-source partner, we can support your complex surgical device throughout the product lifecycle, from design for manufacturability to development to validation and manufacturing.

Injection mold prototyping is the process of developing a tool to expedite a molded part. Spectrum can create your part as close to the intended product design and functionality as possible using your requirements.
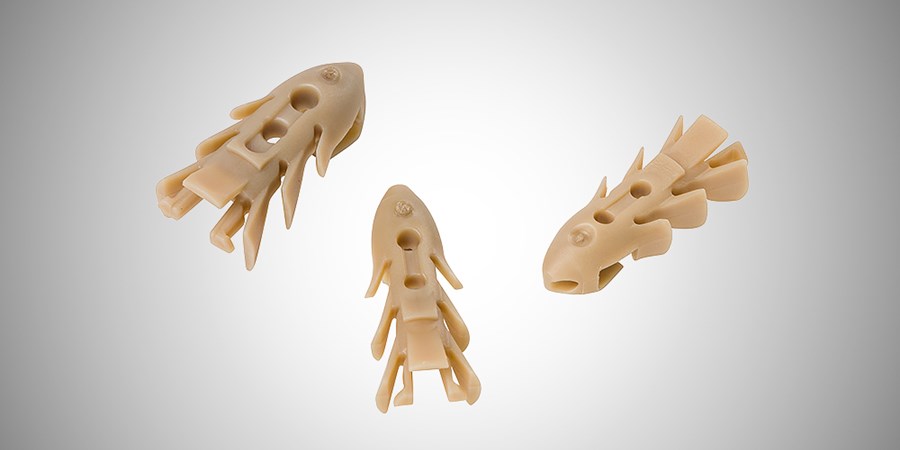
Polyetheretherketone (PEEK) is a high-strength, high-temperature thermoplastic with superior mechanical and chemical properties ideal for injection molding applications.


